What Four Elements Makeup Most Of The Atoms
The abundance of the chemical elements is a measure of the occurrence of the chemical elements relative to all other elements in a given environment. Abundance is measured in one of 3 ways: by the mass-fraction (the same as weight fraction); by the mole-fraction (fraction of atoms by numerical count, or sometimes fraction of molecules in gases); or by the volume-fraction. Volume-fraction is a common affluence measure in mixed gases such as planetary atmospheres, and is similar in value to molecular mole-fraction for gas mixtures at relatively depression densities and pressures, and ideal gas mixtures. Almost abundance values in this article are given equally mass-fractions.
For example, the abundance of oxygen in pure water tin can exist measured in two ways: the mass fraction is about 89%, because that is the fraction of water's mass which is oxygen. However, the mole-fraction is about 33% because only 1 atom of 3 in water, HtwoO, is oxygen. Every bit another example, looking at the mass-fraction abundance of hydrogen and helium in both the Universe as a whole and in the atmospheres of gas-behemothic planets such as Jupiter, it is 74% for hydrogen and 23–25% for helium; while the (atomic) mole-fraction for hydrogen is 92%, and for helium is 8%, in these environments. Changing the given environment to Jupiter's outer atmosphere, where hydrogen is diatomic while helium is not, changes the molecular mole-fraction (fraction of total gas molecules), as well as the fraction of atmosphere by book, of hydrogen to about 86%, and of helium to 13%.[Annotation 1]
The affluence of chemical elements in the universe is dominated by the large amounts of hydrogen and helium which were produced in the Big Bang. Remaining elements, making up but about two% of the universe, were largely produced by supernovae and certain red giant stars. Lithium, glucinium, and boron, despite their low atomic number, are rare considering, although they are produced past nuclear fusion, they are destroyed by other reactions in the stars.[ane] [2] The elements from carbon to atomic number 26 are relatively more abundant in the universe because of the ease of making them in supernova nucleosynthesis. Elements of higher atomic number than iron (element 26) become progressively rarer in the universe, because they increasingly absorb stellar energy in their production. Also, elements with fifty-fifty atomic numbers are mostly more common than their neighbors in the periodic table, due to favorable energetics of germination.
The abundance of elements in the Sun and outer planets is like to that in the universe. Due to solar heating, the elements of Earth and the inner rocky planets of the Solar System have undergone an additional depletion of volatile hydrogen, helium, neon, nitrogen, and carbon (which volatilizes every bit methyl hydride). The crust, mantle, and cadre of the Earth show evidence of chemical segregation plus some sequestration by density. Lighter silicates of aluminium are found in the crust, with more magnesium silicate in the drape, while metallic iron and nickel compose the core. The abundance of elements in specialized environments, such as atmospheres, or oceans, or the human torso, are primarily a product of chemic interactions with the medium in which they reside.
Universe [edit]
| Z | Element | Mass fraction (ppm) |
|---|---|---|
| 1 | Hydrogen | 739,000 |
| 2 | Helium | 240,000 |
| eight | Oxygen | 10,400 |
| six | Carbon | iv,600 |
| 10 | Neon | 1,340 |
| 26 | Fe | 1,090 |
| seven | Nitrogen | 960 |
| 14 | Silicon | 650 |
| 12 | Magnesium | 580 |
| 16 | Sulfur | 440 |
| Full | 999,060 |
The elements – that is, ordinary (baryonic) matter made of protons, neutrons, and electrons, are simply a minor part of the content of the Universe. Cosmological observations propose that just iv.vi% of the universe's energy (including the mass contributed by energy, Eastward = mc ii ↔ m = Eastward /c 2) comprises the visible baryonic matter that constitutes stars, planets, and living beings. The rest is thought to be made up of nighttime energy (68%) and night affair (27%).[4] These are forms of matter and free energy believed to be on the basis of scientific theory and inductive reasoning based on observations, just they have non been directly observed and their nature is not well understood.
Most standard (baryonic) affair is found in intergalactic gas, stars, and interstellar clouds, in the form of atoms or ions (plasma), although it tin can be constitute in degenerate forms in extreme astrophysical settings, such every bit the high densities inside white dwarfs and neutron stars.
Hydrogen is the most abundant element in the Universe; helium is 2nd. All the same, afterwards this, the rank of affluence does not go on to correspond to the atomic number; oxygen has abundance rank 3, but atomic number viii. All others are essentially less common.
The abundance of the lightest elements is well predicted by the standard cosmological model, since they were mostly produced shortly (i.e., within a few hundred seconds) after the Big Bang, in a process known as Big Bang nucleosynthesis. Heavier elements were mostly produced much later, inside of stars.
Hydrogen and helium are estimated to brand up roughly 74% and 24% of all baryonic matter in the universe respectively. Despite comprising only a very small fraction of the universe, the remaining "heavy elements" can profoundly influence astronomical phenomena. Only well-nigh ii% (by mass) of the Galaxy milky way's disk is composed of heavy elements.
These other elements are generated by stellar processes.[5] [6] [vii] In astronomy, a "metal" is any element other than hydrogen or helium. This distinction is significant because hydrogen and helium are the just elements that were produced in significant quantities in the Big Bang. Thus, the metallicity of a galaxy or other object is an indication of stellar action after the Large Blindside.
In general, elements up to iron are made in large stars in the process of condign supernovae. Iron-56 is particularly mutual, since it is the most stable nuclide (in that it has the highest nuclear binding energy per nucleon) and can easily exist made from alpha particles (existence a product of decay of radioactive nickel-56, ultimately made from 14 helium nuclei). Elements heavier than iron are made in energy-absorbing processes in large stars, and their affluence in the universe (and on World) more often than not decreases with increasing diminutive number.

Periodic tabular array showing the cosmological origin of each element
Solar organization [edit]
| Nuclide | A | Mass fraction in parts per million | Cantlet fraction in parts per meg |
|---|---|---|---|
| Hydrogen-1 | one | 705,700 | 909,964 |
| Helium-four | 4 | 275,200 | 88,714 |
| Oxygen-16 | 16 | nine,592 | 477 |
| Carbon-12 | 12 | three,032 | 326 |
| Nitrogen-xiv | 14 | 1,105 | 102 |
| Neon-twenty | 20 | 1,548 | 100 |
| | |||
| Other nuclides: | 3,616 | 172 | |
| Silicon-28 | 28 | 653 | 30 |
| Magnesium-24 | 24 | 513 | 28 |
| Iron-56 | 56 | 1,169 | 27 |
| Sulfur-32 | 32 | 396 | 16 |
| Helium-iii | iii | 35 | 15 |
| Hydrogen-2 | ii | 23 | 15 |
| Neon-22 | 22 | 208 | 12 |
| Magnesium-26 | 26 | 79 | 4 |
| Carbon-13 | thirteen | 37 | four |
| Magnesium-25 | 25 | 69 | four |
| Aluminium-27 | 27 | 58 | 3 |
| Argon-36 | 36 | 77 | 3 |
| Calcium-40 | 40 | 60 | 2 |
| Sodium-23 | 23 | 33 | 2 |
| Atomic number 26-54 | 54 | 72 | ii |
| Silicon-29 | 29 | 34 | ii |
| Nickel-58 | 58 | 49 | 1 |
| Silicon-xxx | 30 | 23 | 1 |
| Iron-57 | 57 | 28 | 1 |
The following graph (note log scale) shows affluence of elements in the Solar Organization. The table shows the twelve most common elements in our galaxy (estimated spectroscopically), every bit measured in parts per million, by mass.[iii] Nearby galaxies that have evolved forth similar lines have a corresponding enrichment of elements heavier than hydrogen and helium. The more than afar galaxies are being viewed equally they appeared in the past, and so their abundances of elements appear closer to the primordial mixture. Since physical laws and processes are compatible throughout the universe, all the same, it is expected that these galaxies volition likewise have evolved like abundances of elements.
The abundance of elements is in keeping with their origin from the Large Bang and nucleosynthesis in a number of progenitor supernova stars. Very arable hydrogen and helium are products of the Big Bang, while the next three elements are rare since they had little fourth dimension to grade in the Big Bang and are non made in stars (they are, however, produced in modest quantities by breakup of heavier elements in interstellar dust, as a result of impact by cosmic rays).
Get-go with carbon, elements have been produced in stars by buildup from alpha particles (helium nuclei), resulting in an alternatingly larger abundance of elements with even atomic numbers (these are too more stable). The upshot of odd-numbered chemical elements more often than not existence more rare in the universe was empirically noticed in 1914, and is known as the Oddo-Harkins rule.

Estimated abundances of the chemical elements in the Solar System (logarithmic scale)
Relation to nuclear binding free energy [edit]
Loose correlations have been observed betwixt estimated elemental abundances in the universe and the nuclear binding energy curve. Roughly speaking, the relative stability of diverse atomic nuclides has exerted a strong influence on the relative abundance of elements formed in the Big Bang, and during the development of the universe thereafter.[9] See the article near nucleosynthesis for an caption of how certain nuclear fusion processes in stars (such as carbon burning, etc.) create the elements heavier than hydrogen and helium.
A further observed peculiarity is the jagged alternation between relative abundance and scarcity of adjacent diminutive numbers in the elemental abundance curve, and a like pattern of energy levels in the nuclear binding energy curve. This alternation is acquired by the higher relative binding energy (corresponding to relative stability) of even atomic numbers compared with odd diminutive numbers and is explained past the Pauli Exclusion Principle.[10] The semi-empirical mass formula (SEMF), too chosen Weizsäcker'south formula or the Bethe-Weizsäcker mass formula, gives a theoretical explanation of the overall shape of the curve of nuclear bounden energy.[11]
World [edit]
The World formed from the same cloud of thing that formed the Sun, simply the planets caused different compositions during the formation and development of the solar organization. In turn, the natural history of the Earth caused parts of this planet to accept differing concentrations of the elements.
The mass of the Earth is approximately 5.97×x 24 kg. In majority, by mass, it is composed mostly of fe (32.i%), oxygen (thirty.ane%), silicon (fifteen.1%), magnesium (13.9%), sulfur (2.9%), nickel (1.eight%), calcium (ane.five%), and aluminium (one.four%); with the remaining 1.ii% consisting of trace amounts of other elements.[12]
The bulk limerick of the Earth by elemental-mass is roughly similar to the gross composition of the solar system, with the major differences being that World is missing a corking deal of the volatile elements hydrogen, helium, neon, and nitrogen, every bit well as carbon which has been lost equally volatile hydrocarbons. The remaining elemental composition is roughly typical of the "rocky" inner planets, which formed in the thermal zone where solar heat drove volatile compounds into space. The Earth retains oxygen every bit the second-largest component of its mass (and largest atomic-fraction), mainly from this element being retained in silicate minerals which have a very loftier melting bespeak and low vapor pressure level.
| Diminutive Number | Name | Symbol | Mass fraction (ppm) | Atomic fraction (ppb)[xiii] |
|---|---|---|---|---|
| 8 | oxygen | O | 297000 | 482,000,000 |
| 12 | magnesium | Mg | 154000 | 164,000,000 |
| 14 | silicon | Si | 161000 | 150,000,000 |
| 26 | iron | Iron | 319000 | 148,000,000 |
| 13 | aluminium | Al | 15900 | fifteen,300,000 |
| 20 | calcium | Ca | 17100 | xi,100,000 |
| 28 | nickel | Ni | 18220 | 8,010,000 |
| 1 | hydrogen | H | 260 | 6,700,000 |
| 16 | sulfur | S | 6350 | 5,150,000 |
| 24 | chromium | Cr | 4700 | two,300,000 |
| 11 | sodium | Na | 1800 | ii,000,000 |
| 6 | carbon | C | 730 | 1,600,000 |
| 15 | phosphorus | P | 1210 | 1,020,000 |
| 25 | manganese | Mn | 1700 | 800,000 |
| 22 | titanium | Ti | 810 | 440,000 |
| 27 | cobalt | Co | 880 | 390,000 |
| 19 | potassium | K | 160 | 110,000 |
| 17 | chlorine | Cl | 76 | 56,000 |
| 23 | vanadium | Five | 105 | 53,600 |
| seven | nitrogen | N | 25 | 46,000 |
| 29 | copper | Cu | 60 | 25,000 |
| thirty | zinc | Zn | forty | 16,000 |
| 9 | fluorine | F | ten | 14,000 |
| 21 | scandium | Sc | 11 | 6,300 |
| 3 | lithium | Li | one.10 | 4,100 |
| 38 | strontium | Sr | thirteen | 3,900 |
| 32 | germanium | Ge | vii.00 | 2,500 |
| 40 | zirconium | Zr | 7.x | 2,000 |
| 31 | gallium | Ga | three.00 | 1,000 |
| 34 | selenium | Se | two.70 | 890 |
| 56 | barium | Ba | 4.50 | 850 |
| 39 | yttrium | Y | 2.ninety | 850 |
| 33 | arsenic | As | 1.lxx | 590 |
| v | boron | B | 0.20 | 480 |
| 42 | molybdenum | Mo | 1.70 | 460 |
| 44 | ruthenium | Ru | 1.30 | 330 |
| 78 | platinum | Pt | i.90 | 250 |
| 46 | palladium | Pd | i.00 | 240 |
| 58 | cerium | Ce | i.13 | 210 |
| sixty | neodymium | Nd | 0.84 | 150 |
| 4 | beryllium | Be | 0.05 | 140 |
| 41 | niobium | Nb | 0.44 | 120 |
| 76 | osmium | Os | 0.ninety | 120 |
| 77 | iridium | Ir | 0.90 | 120 |
| 37 | rubidium | Rb | 0.twoscore | 120 |
| 35 | bromine | Br | 0.30 | 97 |
| 57 | lanthanum | La | 0.44 | 82 |
| 66 | dysprosium | Dy | 0.46 | 74 |
| 64 | gadolinium | Gd | 0.37 | 61 |
| 52 | tellurium | Te | 0.30 | 61 |
| 45 | rhodium | Rh | 0.24 | 61 |
| 50 | tin | Sn | 0.25 | 55 |
| 62 | samarium | Sm | 0.27 | 47 |
| 68 | erbium | Er | 0.30 | 47 |
| 70 | ytterbium | Yb | 0.30 | 45 |
| 59 | praseodymium | Pr | 0.17 | 31 |
| 82 | lead | Pb | 0.23 | 29 |
| 72 | hafnium | Hf | 0.19 | 28 |
| 74 | tungsten | W | 0.17 | 24 |
| 79 | aureate | Au | 0.xvi | 21 |
| 48 | cadmium | Cd | 0.08 | eighteen |
| 63 | europium | Eu | 0.x | 17 |
| 67 | holmium | Ho | 0.10 | 16 |
| 47 | silver | Ag | 0.05 | 12 |
| 65 | terbium | Tb | 0.07 | xi |
| 51 | antimony | Sb | 0.05 | 11 |
| 75 | rhenium | Re | 0.08 | 10 |
| 53 | iodine | I | 0.05 | x |
| 69 | thulium | Tm | 0.05 | 7 |
| 55 | cesium | Cs | 0.04 | 7 |
| 71 | lutetium | Lu | 0.05 | seven |
| 90 | thorium | Th | 0.06 | half-dozen |
| 73 | tantalum | Ta | 0.03 | 4 |
| 80 | mercury | Hg | 0.02 | 3 |
| 92 | uranium | U | 0.02 | ii |
| 49 | indium | In | 0.01 | two |
| 81 | thallium | Tl | 0.01 | 2 |
| 83 | bismuth | Bi | 0.01 | ane |
Crust [edit]
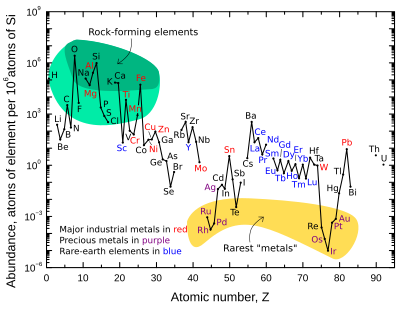
Abundance (atom fraction) of the chemical elements in Earth's upper continental crust equally a role of diminutive number. The rarest elements in the crust (shown in yellow) are rare due to a combination of factors: all but one are the densest siderophiles (fe-loving) elements in the Goldschmidt nomenclature, meaning they have a trend to mix well with metallic atomic number 26, depleting them by beingness relocated deeper into the Globe's core. Their abundance in meteoroids is higher. Additionally, tellurium has been depleted by preaccretional sorting in the nebula via formation of volatile hydrogen telluride.[14]
The mass-abundance of the ix most abundant elements in the World'due south crust is approximately: oxygen 46%, silicon 28%, aluminium eight.3%, iron 5.half dozen%, calcium 4.2%, sodium 2.v%, magnesium 2.four%, potassium ii.0%, and titanium 0.61%. Other elements occur at less than 0.fifteen%. For a consummate list, encounter affluence of elements in Earth'southward chaff.
The graph at right illustrates the relative diminutive-abundance of the chemical elements in Earth'south upper continental crust—the function that is relatively accessible for measurements and estimation.
Many of the elements shown in the graph are classified into (partially overlapping) categories:
- rock-forming elements (major elements in green field, and minor elements in light light-green field);
- rare globe elements (lanthanides (La–Lu), Sc, and Y; labeled in bluish);
- major industrial metals (global product >~3×ten7 kg/year; labeled in reddish);
- precious metals (labeled in imperial);
- the nine rarest "metals" – the vi platinum group elements plus Au, Re, and Te (a metalloid) – in the yellow field. These are rare in the crust from being soluble in iron and thus full-bodied in the Earth's core. Tellurium is the single most depleted element in the silicate Globe relative to cosmic abundance, considering in addition to being concentrated equally dense chalcogenides in the cadre it was severely depleted by preaccretional sorting in the nebula as volatile hydrogen telluride.[fourteen]
Annotation that there are two breaks where the unstable (radioactive) elements technetium (atomic number 43) and promethium (diminutive number 61) would be. These elements are surrounded by stable elements, nevertheless their about stable isotopes have relatively brusque half lives (~4 million years and ~18 years respectively). These are thus extremely rare, since any primordial initial fractions of these in pre-Solar System materials have long since decayed. These two elements are now but produced naturally through the spontaneous fission of very heavy radioactive elements (for example, uranium, thorium, or the trace amounts of plutonium that be in uranium ores), or past the interaction of certain other elements with cosmic rays. Both technetium and promethium have been identified spectroscopically in the atmospheres of stars, where they are produced by ongoing nucleosynthetic processes.
There are also breaks in the affluence graph where the half dozen noble gases would be, since they are not chemically bound in the Earth'south crust, and they are only generated in the chaff past disuse chains from radioactive elements, and are therefore extremely rare there.
The eight naturally occurring very rare, highly radioactive elements (polonium, astatine, francium, radium, actinium, protactinium, neptunium, and plutonium) are not included, since any of these elements that were present at the germination of the Earth have decayed away eons ago, and their quantity today is negligible and is only produced from the radioactivity of uranium and thorium.
Oxygen and silicon are notably the virtually common elements in the crust. On Earth and in rocky planets in general, silicon and oxygen are far more common than their cosmic abundance. The reason is that they combine with each other to course silicate minerals.[fourteen] Other cosmically-common elements such as hydrogen, carbon and nitrogen form volatile compounds such as ammonia and marsh gas that easily boil away into space from the heat of planetary formation and/or the Lord's day'due south light.
Rare-world elements [edit]
"Rare" earth elements is a historical misnomer. The persistence of the term reflects unfamiliarity rather than true rarity. The more abundant rare globe elements are similarly full-bodied in the chaff compared to commonplace industrial metals such as chromium, nickel, copper, zinc, molybdenum, tin, tungsten, or lead. The two least abundant rare globe elements (thulium and lutetium) are well-nigh 200 times more common than gold. However, in contrast to the ordinary base of operations and precious metals, rare earth elements have very trivial trend to get concentrated in exploitable ore deposits. Consequently, most of the world's supply of rare globe elements comes from only a handful of sources. Furthermore, the rare earth metals are all quite chemically similar to each other, and they are thus quite hard to separate into quantities of the pure elements.
Differences in abundances of individual rare world elements in the upper continental crust of the Earth stand for the superposition of ii effects, 1 nuclear and ane geochemical. Commencement, the rare earth elements with fifty-fifty atomic numbers (58Ce, 60Nd, ...) accept greater cosmic and terrestrial abundances than the adjacent rare earth elements with odd atomic numbers (57La, 59Pr, ...). Second, the lighter rare earth elements are more incompatible (because they have larger ionic radii) and therefore more than strongly concentrated in the continental crust than the heavier rare earth elements. In most rare earth ore deposits, the beginning four rare earth elements – lanthanum, cerium, praseodymium, and neodymium – constitute 80% to 99% of the total amount of rare earth metal that tin can be plant in the ore.
Mantle [edit]
The mass-abundance of the 8 near arable elements in the Globe's pall is approximately: oxygen 44.iii%, magnesium 22.iii%, silicon 21.3%, fe half-dozen.32%, calcium 2.48%, aluminium 2.29%, nickle 0.nineteen%.[15]
Cadre [edit]
Due to mass segregation, the core of the Earth is believed to be primarily composed of fe (88.8%), with smaller amounts of nickel (5.8%), sulfur (4.5%), and less than one% trace elements.[12]
Ocean [edit]
The most arable elements in the ocean past proportion of mass in percent are oxygen (85.84%), hydrogen (10.82%), chlorine (1.94%), sodium (1.08%), magnesium (0.13%), sulfur (0.09%), calcium (0.04%), potassium (0.04%), bromine (0.007%), carbon (0.003%), and boron (0.0004%).
Atmosphere [edit]
The society of elements by volume-fraction (which is approximately molecular mole-fraction) in the temper is nitrogen (78.1%), oxygen (20.9%),[xvi] argon (0.96%), followed by (in uncertain society) carbon and hydrogen because water vapor and carbon dioxide, which represent most of these two elements in the air, are variable components. Sulfur, phosphorus, and all other elements are nowadays in significantly lower proportions.
Co-ordinate to the affluence curve graph, argon, a significant if not major component of the atmosphere, does not announced in the crust at all. This is because the atmosphere has a far smaller mass than the chaff, then argon remaining in the crust contributes footling to mass-fraction in that location, while at the same time buildup of argon in the atmosphere has become large plenty to be significant.
Urban soils [edit]
For a complete list of the abundance of elements in urban soils, see Abundances of the elements (data page)#Urban soils.
Human body [edit]
| Element | Proportion (by mass) |
|---|---|
| Oxygen | 65 |
| Carbon | eighteen |
| Hydrogen | 10 |
| Nitrogen | 3 |
| Calcium | ane.v |
| Phosphorus | ane.2 |
| Potassium | 0.2 |
| Sulfur | 0.2 |
| Chlorine | 0.2 |
| Sodium | 0.i |
| Magnesium | 0.05 |
| Iron | < 0.05 |
| Cobalt | < 0.05 |
| Copper | < 0.05 |
| Zinc | < 0.05 |
| Iodine | < 0.05 |
| Selenium | < 0.01 |
By mass, human being cells consist of 65–90% h2o (H2O), and a pregnant portion of the residuum is composed of carbon-containing organic molecules. Oxygen therefore contributes a majority of a homo body's mass, followed past carbon. Near 99% of the mass of the human body is made up of six elements: hydrogen (H), carbon (C), nitrogen (Northward), oxygen (O), calcium (Ca), and phosphorus (P) . The side by side 0.75% is fabricated up of the next five elements: potassium (Thou), sulfur (S), chlorine (Cl), sodium (Na), and magnesium (Mg). Only 17 elements are known for certain to be necessary to human life, with one additional element (fluorine) thought to exist helpful for tooth enamel forcefulness. A few more trace elements may play some role in the health of mammals. Boron and silicon are notably necessary for plants but have uncertain roles in animals. The elements aluminium and silicon, although very mutual in the earth's crust, are clearly rare in the human body.[17]
Below is a periodic table highlighting nutritional elements.[18]
| Nutritional elements in the periodic table[xix] | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H | He | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Li | Exist | B | C | N | O | F | Ne | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Na | Mg | Al | Si | P | Due south | Cl | Ar | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| K | Ca | Sc | Ti | V | Cr | Mn | Fe | Co | Ni | Cu | Zn | Ga | Ge | As | Se | Br | Kr | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rb | Sr | Y | Zr | Nb | Mo | Tc | Ru | Rh | Pd | Ag | Cd | In | Sn | Sb | Te | I | Xe | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cs | Ba | * | Lu | Hf | Ta | Due west | Re | Os | Ir | Pt | Au | Hg | Tl | Pb | Bi | Po | At | Rn | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fr | Ra | ** | Lr | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * | La | Ce | Pr | Nd | Pm | Sm | Eu | Gd | Tb | Dy | Ho | Er | Tm | Yb | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ** | Air-conditioning | Th | Pa | U | Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Legend:
Quantity elements
Accounted essential trace chemical element by U.S., not by European Union
Suggested function from impecuniousness effects or agile metabolic handling, simply no clearly-identified biochemical role in humans
Express circumstantial bear witness for trace benefits or biological action in mammals
No bear witness for biological activity in mammals, but essential in some lower organisms.
(In the case of lanthanum, the definition of an essential food every bit being indispensable and irreplaceable is not completely applicable due to the farthermost similarity of the lanthanides. The stable early lanthanides up to Sm are known to stimulate the growth of various lanthanide-using organisms.)[20]
See also [edit]
- Abundances of the elements (data page)
- Abundance of elements in Earth's crust
- Natural affluence (isotopic abundance)
- Goldschmidt nomenclature
- Primordial nuclide
- Radiative levitation
- List of information references for chemical elements
References [edit]
Footnotes [edit]
- ^ Vangioni-Flam, Elisabeth; Cassé, Michel (2012). Spite, Monique (ed.). Galaxy Development: Connecting the Distant Universe with the Local Fossil Record. Springer Science & Business organisation Media. pp. 77–86. ISBN978-9401142137.
- ^ Trimble, Virginia (1996). "The Origin and Evolution of the Chemical Elements". In Malkan, Matthew A.; Zuckerman, Ben (eds.). The origin and development of the universe. Sudbury, Mass.: Jones and Bartlett Publishers. p. 101. ISBN0-7637-0030-iv.
- ^ a b Croswell, Ken (February 1996). Alchemy of the Heavens. Anchor. ISBN0-385-47214-5. Archived from the original on 2011-05-13.
- ^ What is Dark Energy? Archived 2016-01-15 at the Wayback Machine, Space.com, 1 May 2013.
- ^ Suess, Hans; Urey, Harold (1956). "Abundances of the Elements". Reviews of Mod Physics. 28 (1): 53. Bibcode:1956RvMP...28...53S. doi:x.1103/RevModPhys.28.53.
- ^ Cameron, A. G. Westward. (1973). "Abundances of the elements in the solar system". Space Scientific discipline Reviews. 15 (1): 121. Bibcode:1973SSRv...fifteen..121C. doi:10.1007/BF00172440. S2CID 120201972.
- ^ Anders, E.; Ebihara, M. (1982). "Solar-system abundances of the elements". Geochimica et Cosmochimica Acta. 46 (11): 2363. Bibcode:1982GeCoA..46.2363A. doi:10.1016/0016-7037(82)90208-3.
- ^ Arnett, David (1996). Supernovae and Nucleosynthesis (Start ed.). Princeton, New Jersey: Princeton Academy Press. p. 11. ISBN0-691-01147-viii. OCLC 33162440.
- ^ Bell, Jerry A.; GenChem Editorial/Writing Team (2005). "Chapter three: Origin of Atoms". Chemistry: a project of the American Chemical Club. New York [u.a.]: Freeman. pp. 191–193. ISBN978-0-7167-3126-9.
Correlations between abundance and nuclear binding energy [Subsection title]
- ^ Bong, Jerry A.; GenChem Editorial/Writing Team (2005). "Chapter 3: Origin of Atoms". Chemical science: a project of the American Chemical Social club. New York [u.a.]: Freeman. p. 192. ISBN978-0-7167-3126-9.
The higher abundance of elements with even diminutive numbers [Subsection title]
- ^ Bailey, David. "Semi-empirical Nuclear Mass Formula". PHY357: Strings & Binding Energy. University of Toronto. Archived from the original on 2011-07-24. Retrieved 2011-03-31 .
- ^ a b Morgan, J. W.; Anders, Eastward. (1980). "Chemical composition of World, Venus, and Mercury". Proceedings of the National University of Sciences. 77 (12): 6973–6977. Bibcode:1980PNAS...77.6973M. doi:10.1073/pnas.77.12.6973. PMC350422. PMID 16592930.
- ^ a b William F McDonough The composition of the Earth. convulse.mit.edu, archived by the Internet Annal Wayback Machine.
- ^ a b c Anderson, Don L.; 'Chemical Composition of the Mantle' in Theory of the Globe, pp. 147–175 ISBN 0865421234
- ^ Wang, Haiyang Due south.; Lineweaver, Charles H.; Ireland, Trevor R. (2018-01-01). "The elemental abundances (with uncertainties) of the most Earth-like planet". Icarus. 299: 460–474. doi:x.1016/j.icarus.2017.08.024. ISSN 0019-1035.
- ^ Zimmer, Carl (three October 2013). "Earth'southward Oxygen: A Mystery Easy to Take for Granted". The New York Times. Archived from the original on three October 2013. Retrieved iii Oct 2013.
- ^ Table data from Chang, Raymond (2007). Chemical science (Ninth ed.). McGraw-Hill. p. 52. ISBN978-0-07-110595-8.
- ^ Nielsen, Forrest H. (1998). "Ultratrace minerals.". In Maurice E. Shils; James A. Olsen; Moshe Shine; A. Catharine Ross (eds.). Modern nutrition in health and disease. Baltimore: Lippincott Williams & Wilkins. pp. 283–303. hdl:10113/46493. ISBN978-0683307696.
- ^ Ultratrace minerals. Authors: Nielsen, Forrest H. USDA, ARS Source: Modernistic nutrition in wellness and disease / editors, Maurice East. Shils ... et al. Baltimore: Williams & Wilkins, c1999., p. 283-303. Issue Date: 1999 URI: [1]
- ^ Daumann, Lena J. (25 April 2019). "Essential and Ubiquitous: The Emergence of Lanthanide Metallobiochemistry". Angewandte Chemie International Edition. doi:10.1002/anie.201904090. Retrieved 15 June 2019.
Notes [edit]
- ^ Below Jupiter'south outer atmosphere, volume fractions are significantly different from mole fractions due to high temperatures (ionization and disproportionation) and high density where the Ideal Gas Police is inapplicable.
Notations [edit]
- "Rare Earth Elements—Critical Resources for High Technology | USGS Fact Sheet 087-02". geopubs.wr.usgs.gov.
- "Imagine the Universe! Dictionary". 3 December 2003. Archived from the original on 3 Dec 2003.
External links [edit]
- Listing of elements in club of abundance in the Earth's chaff (only correct for the twenty most mutual elements)
- Cosmic abundance of the elements and nucleosynthesis
- WebElements.com Lists of elemental abundances for the Universe, Sun, meteorites, Earth, ocean, streamwater, etc.
Source: https://en.wikipedia.org/wiki/Abundance_of_the_chemical_elements
Posted by: pritchardtrieste.blogspot.com

0 Response to "What Four Elements Makeup Most Of The Atoms"
Post a Comment